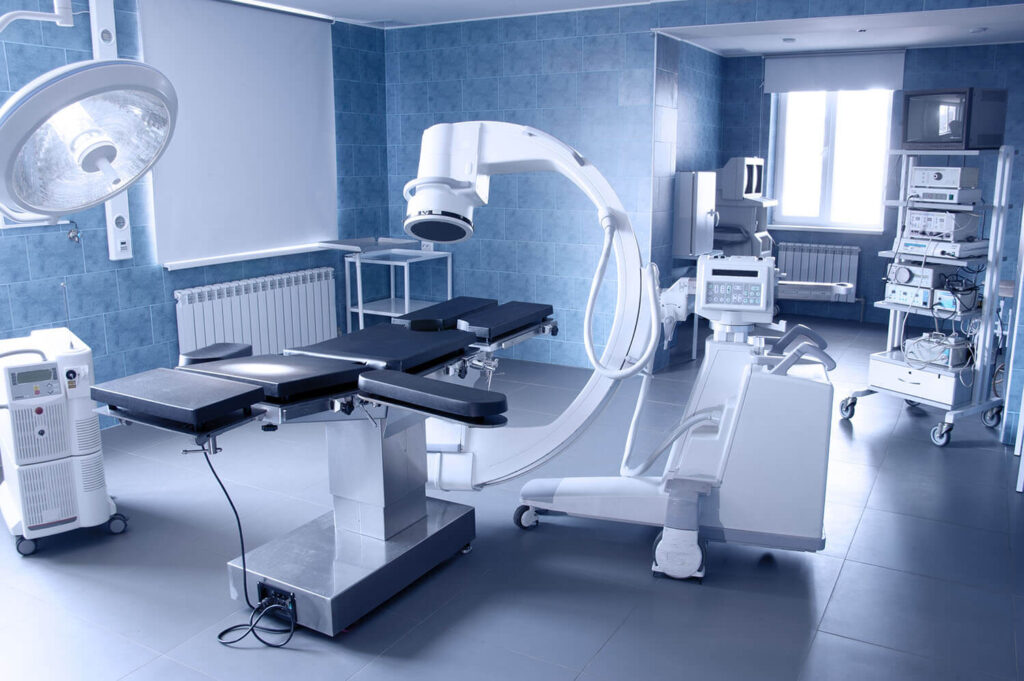
ISO 13485 Medical Devices Manufacturing Implementation (QMS)
Whether you’re in an elevator or at the doctor’s office, one of the first items you’ll definitely find is some form of credential hanging on the wall. Certifications provide guarantees to customers that the product or services they are using can work or provide them with the outcomes they want. Certifications are important when you’re in the medical device business because there are many guidelines and regulations that you have to conform to when you sell goods in this area. However, one of the most critical norms is ISO 13485. Not only does the company have to be accredited to have this quality control level for the medical devices sector, but it is also necessary to ensure that the whole supply chain always follows such rigorous demands.
First A Definition of Medical Devices:
“Health equipment varies from basic depressors of the tongue and bedpans to sophisticated programmable pacemakers with microchip technology and laser surgical instruments. Additionally, medical instruments include in vitro diagnostic items, such as general-purpose laboratory equipment, reagents, and test kits, which could include monoclonal antibody technologies. Some electronic radiation emitted computer-application goods and statements follow the medical system concept. Examples include ultrasound diagnostics devices, x-ray machines, and lasers for medical purposes. If a product is labeled, promoted or used in a manner that meets the following definition in section 201(h) of the Federal Food Drug & Cosmetic (FD&C) Act, it is regulated as a medical device by the Food and Drug Administration (FDA) and subject to regulatory pre-marketing and post-marketing controls.
A medical device is:
An instrument, a tool, an implement, a computer, an implant, an in vitro reagent, and/or another specific or connected object, including one component part or an accessory that is:
Any medical device used in the U.S., whether made in the U.S. or not, must be approved in compliance with FDA guidelines in 21 CFR 820 which have been modified for medical devices as ISO 13485 by ISO Standards. ISO 13485:2016 is the latest to be used. This standard follows ISO 9001:2008 prescribed Quality Management systems.
So what is ISO 13485 all about?
ISO 13485 is the globally recognized standard in the medical device sector for quality control programs. It defines specifications for a quality control program where a company wants to show the capacity to deliver medical equipment, and associated facilities reliably satisfy consumer expectations and regulatory criteria that exist. It’s designed as well as intended to be used by an organization for medical device development and design, manufacture, installation, servicing, as well as sales.
ISO 13485 specifically seeks to promote harmonized legislative standards for quality control programs for medical devices. ISO 13485 has a specification and is independent. It is by and large based upon the ISO 9001 framework, but has some specific requirements for all medical devices including risk analysis, importantly sterile manufacturing as well as traceability. Organizations that are ISO 13485 accredited can not assert compliance with ISO 9001.
Why is a quality management system important to you for your medical devices?
Being a professional certified ISO 13485 shows your determination in offering your company as well as customers quality products and services. Achieving the important knowledge and skills to effectively operate an ISO 13485 framework which demonstrates your continued commitment to helping ensure continued improvement and improved work processes in your company.
Benefits of ISO 13485 Quality Management System for medical devices
When you contract ISO Pros to become a professional company certified in ISO 13485, you’ll be able to:
- Increase the number of employment opportunities
- Establish new market ties
- Earn a competitive edge
- Growing efficiency and lowering costs
- Provide expertise to improve workflows
- Provide healthcare tools that are healthier and more effective
- Raises customer satisfaction
This is applicable to all companies engaged in planning, developing, integrating, and servicing medical equipment and associated facilities.
The new version of the internationally recognized specification, recently updated to ISO 13485:2016, provides a framework for fulfilling the requirements of a quality management system for medical devices that seeks to address the entire lifecycle of a medical device.
This includes the below:
System for Controlling Consistency
In accordance with ISO 9001 standards, ISO 13485 follows the structure for implementing a quality management system and criteria for reporting procedures for the highly regulated medical device industry. Expands the Standard terms to cover areas such as clinical evaluation, applications, families of medical devices, post-market monitoring, manufacturers, and importers.
Responsibility of Management
Top management is at the center of the success of the organization and is therefore required to establish quality processes and goals and to perform evaluations to ensure that the business reaches the expectations of the customer.
Managing Resources
Implement the right people and infrastructure to foster a positive work environment that sets the organization to success, only applicable where relevant.
Product Realisation
The Framework addresses the criteria that the company needs to fulfill in the planning, design, and development, procurement, and control of the medical device – only appropriate where necessary.
Measuring, Analyzing and Improving
Organizations will need to monitor and measure the performance of business processes (only applicable where relevant), including meeting customer expectations. Internal audits allow for continuous enhancement and corrective action.
Which position does the customer play in all this?
Of course, the consumer is always king, and if you can’t draw and please your customers sufficiently, all the laws in the world will make no difference to a market! Many companies are able to design and promote products, but the consistent quality and customer satisfaction attention are what bring you long-term market success. In reality, customer service is a primary priority of the ISO 13485 standard. By certifying to an ISO management system standard, you can improve yourself as an excellent and consistent supplier to all your customers. When the value of conformity and loyalty increases, a range of clients who do not partner with non-ISO accredited organizations would make your company more attractive.
A number of companies consider that it doesn’t matter how strong their quality is, consumers, require more reassurance if they want approval by their suppliers. In fact, several companies consider that it’s worth losing one deal, as it will offset the expense of adopting effective business practices and becoming accredited. If a management system has been well documented as well as implemented, with the increased knowledge that your great business is always in safe hands and operates as it should be, you might even enjoy life a little more.
Eligibility to receive ISO 13485 Certification
ISO 13485 is issued to organizations involved directly or indirectly in one or more phases of a medical device’s life-cycle. Any company that supervises one or more of such processes engages directly in the manufacturing of a medical device:
- Researching, designing, and development
- Manufacturing and production
- Store, distribute, install, and maintenance
Indirect participation includes supplying the goods and facilities directly to the businesses engaged in the manufacturing of medical equipment. Consequently, contractors and suppliers offering calibration, sterilization, and the maintenance of raw materials, components, and services to sustain the life cycle of medical devices and are also eligible for ISO 13485 certification.
In order to achieve ISO 13485, an organization must define its position and obligations to the medical device industry in order to meet a series of regulatory criteria established by the ISO for that particular function. To comply with ISO 13485, for example, medical product suppliers and retailers would need to incorporate specific collections of steps into their quality control framework.
Who issues certificates according to ISO?
An approved institution for quality assurance verifies and then provides a certificate that a client complies with the specific quality control standards provided by the standardization body.
Consider it for the EU market as well
ISO 13485 also acts as the QMS standard for obtaining a CE mark on medical devices, accepted under European regulations. CE branding reflects the argument of the supplier that their drug complies with the applicable EU Directives on Medical Devices and allows for sale within the European Union. The selling of pharmaceutical goods within the European Union is legally necessary.
If your medical system is spreading into European markets and you need the CE label, you need to start converting or being accredited according to ISO 13485.
Why use ISO Pros for your certification
Our technical professionals have in-depth knowledge and expertise with all types of medical devices. We are now a major global rating agency for quality control programs, including management systems that are important in the manufacturing of medical equipment. Our audit team is composed of professionals with the skills and expertise needed to accurately determine the management system’s compliance and is guided by a lead auditor who will monitor the audit and any notifications required to change management. Finally, the details you need to appreciate your vulnerability to non-compliance problems would be given through our on-site or off-site Professional Data Review. This rare mix of expertise allows ISO Pros to be best positioned to fulfill the needs of producers of medical devices requiring ISO 13485 approval or its maintenance.
ISO Pros offers a wide spectrum of research, inspection, and evaluation facilities to medical device suppliers, helping them mitigate hazards and ensuring and supporting the health as well as the welfare of customers, consumers, and, where necessary, other individuals. We have a global network of over many committed medical health care providers consisting of established scientists, physicians in their fields who are regarded as experts. These capabilities make ISO Pros the preferred single source for compliance with regulations regarding medical devices worldwide.
What is the objective of the certification
The main objective is to provide on the international market a harmonized model for quality management system requirements, since different countries may have different standards. While it remains a separate text, in general, ISO 13485 is harmonized with ISO 9001.
It includes some specific medical device requirements, with emphasis on;
Compliance with ISO 13485 is often seen as the first step towards fulfilling the regulatory requirements of the European Union as well.
We have no preconceived ideas on how you should adapt quality management to your company. It is your business and you know how best to implement the requirements of the relevant norms. Our role is to assess whether your management system actually complies with the relevant standard irrespective of how you do that! So contact ISO Pros today for all your ISO 13485 certification.
FAQ: The response to commonly asked questions
What do you intend to do when performing audits?
If you wish to sell a medical product on the market, an examination by a notified entity is needed. An exemption is for products classified as Class I.
How long does one take for an audit?
This depends, inter alia, on the scale of your company. As a rule, 5-employee companies must expect 2.5 days, 10 employees 3 days, 50 employees 6 days, and 100 employees 8 days.
Who is permitted to do audits?
That depends on the objective of the audit. If you are looking for ISO 13485 qualification, informed bodies are approved exclusively.
What needs to be addressed in the form of the audits?
In respect to an evaluation of ISO 13485, you will provide a recorded quality control program, and need to stick to it. For example, this means you must have created and manufactured a product consistent with the requirements of the standard for at least the most part. Hence, the technical documentation of a medical device must be (substantially) accurate. Auditors would like to analyze how the QM-system satisfies the standard criteria and whether you comply with the QM-system.